ISO 13485:2016 Medical Device MDQMS
Things You Need to Know
ISO 13485:2016 Medical Device MDQMS
ISO 13485:2016 Medical Device MDQMS
-
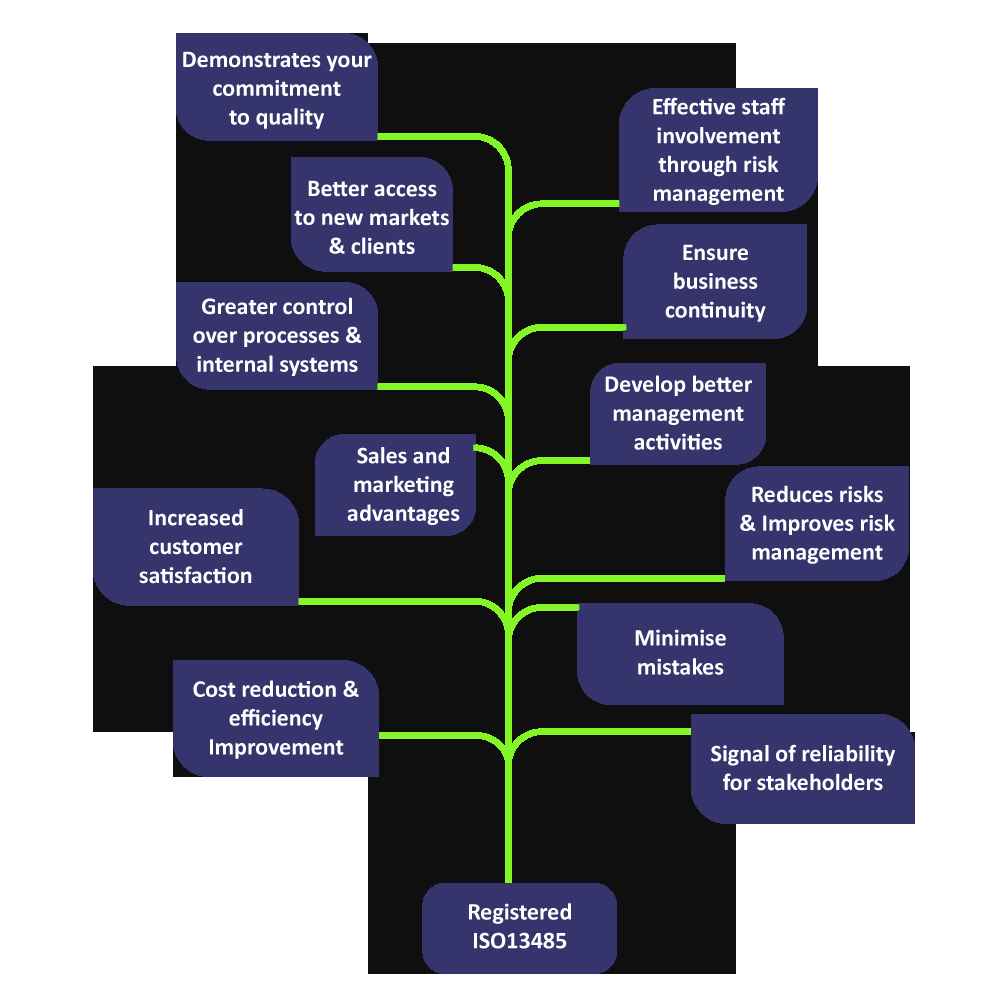
ISO 13485 improves the performance of the products & processes of a medical device company.
ISO 13485 certificate is universally recognized & it covers regulatory & statutory norms of most of the countries, your product gets expanded market access.
ISO 13485 works as a brand ambassador of your medical device hence your sales & marketing cost will reduce.
ISO 13485 certification gives faith to your customers that the device is safe for use & it meets the requirements of most of the recognized technical standards related to the medical device.














